How can I identify meso compounds?
1 Answer
You look for an internal plane of symmetry.
Explanation:
A meso compound must have
- Two or more chiral centres and
- An internal plane of symmetry
A plane of symmetry is any plane that cuts through the molecule and makes one side a perfect reflection of the other.
When you look for a plane of symmetry, you must put the molecule in the most symmetric conformation possible.
This means that you can look for symmetry in cyclic structures that are drawn flat and in alkanes that are eclipsed.
The two structures on the right are meso compounds, because the top half is a mirror image of the bottom half.
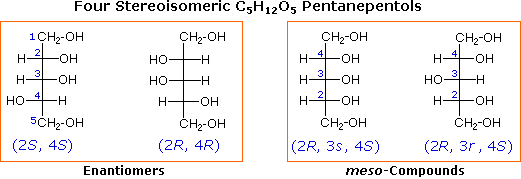
Note that the mirror planes can bisect atoms.
If you know the R,S notation, you can use this procedure.
Identify the R and S stereochemistry at each chiral centre.
If 2 chiral centres, R,S = S,R = meso.
If 3 chiral centres, R,R,S = R,S,S = meso, and S,R,R = S,S,R = meso. The configuration of the middle carbon doesn't matter, because the plane of symmetry will bisect that atom.
If 4 chiral centres, R,S,S,R = S,R,R,S = meso, and R,R,S,S = S,S,R,R = meso.
This method is often easier than trying to look for mirror images by rotating molecules in your mind or by redrawing structures in various orientations.


