What mode of hybridization is associated with each of the five common electron domain geometries?
1 Answer
Jun 6, 2015
The hybridization uses first
We can classify the electron geometries according to the
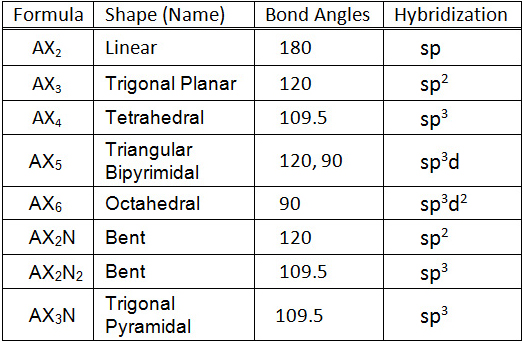
#"AX"_2# = linear =#sp# hybridization#"AX"_3# = trigonal planar =#sp^2# hybridization#"AX"_4# = tetrahedral =#sp^3# hybridization#"AX"_5# = trigonal bipyramidal =#sp^3d# hybridization#"AX"_6# = octahedral =#sp^3d^2# hybridization